The AMV Analytical Method Validation is a software designed for the statistical evaluation of validation characteristics according to ICH Q2 recommendations. The program consists of 7 modules.

Basic module functions:
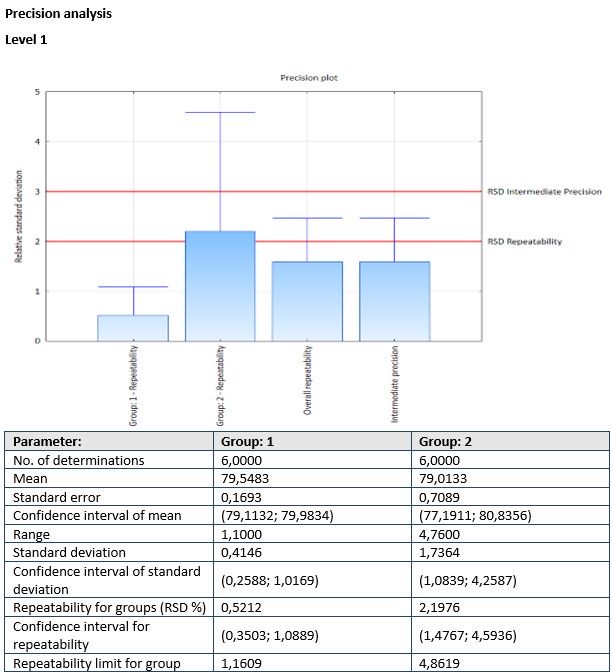
- Module PRECISION is used to perform the statistical analysis of method precision (repeatability and/or intermediate precision) for different concentration levels and groups.
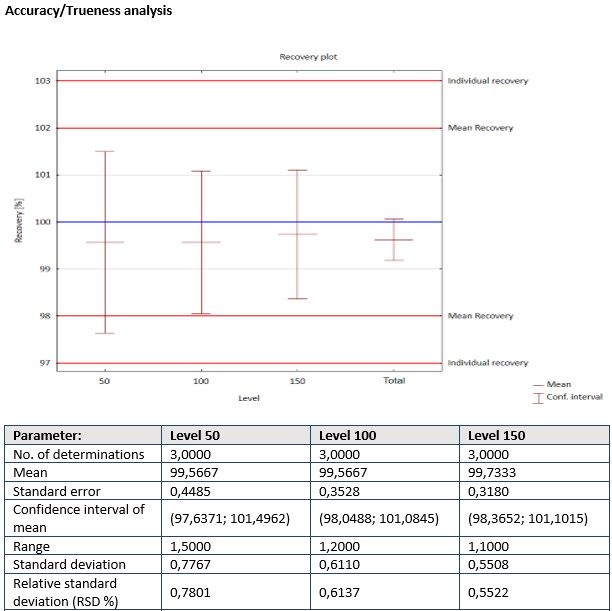
- Module ACCURACY/TRUENESS is used to perform the statistical analysis of method accuracy/trueness for different concentration levels and groups.
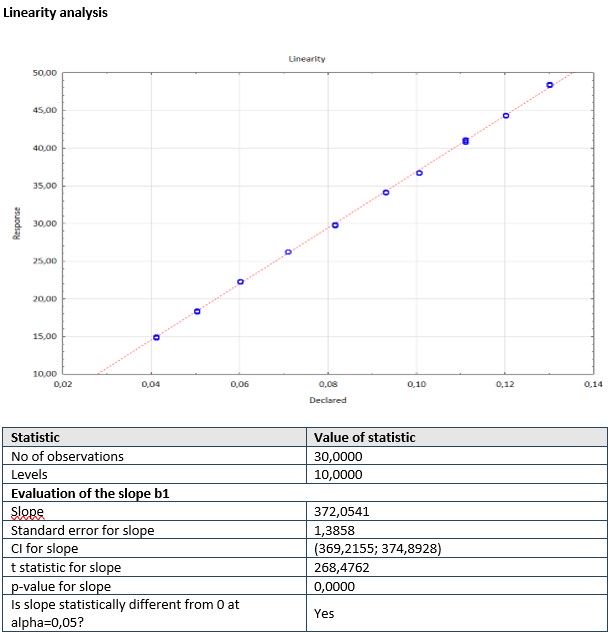
- Module LINEARITY/CALIBRATION is used to perform the statistical analysis of linearity/calibration based on the declared and response value of the instrument or the method.
- Module LOQ/LOD is used to perform the statistical analysis of the limit of detection and quantitation based on the value of the blank or calibration curve.
- Module GROUPS COMPARISON is used to perform the statistical comparative analysis of two groups of results to evaluate method transfer between laboratories or to compare two alternative methods.
- Module REGRESSION CURVE COMPARISON is used to perform the statistical comparative analysis of regression lines.
- Module ACCURACY acc. to ISO 5725 is used to perform the statistical analysis of accuracy (trueness and precision) and estimation of the uncertainty of measurement based on the inter laboratory study.
The application allows:
- selection of an appropriate statistical method for evaluation of validation characteristics,
- standardization of the performed statistical analyzes and interpretation of the results,
- automatic report generation in tabular and/or graphical form.
All you need is to enter the data and press a button to get a full report in the form of a Microsoft Word or PDF document. The report includes the results of the statistical analyses and comments to facilitate the interpretation of the obtained results. The methods used in the module comply to the recommendations of the EU EMA and the US FDA.
The software is validated according to cGxP requirements.